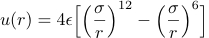 ,
,
Introduction
The exact form of the interparticle potential u(r) for electrically neutral molecules and atoms has to be determined by a first principles quantum mechanical calculation. Such a calculation is very difficult, and for many purposes it is sufficient to choose a simple phenomenological form for u(r). The most important features of u(r) are a strong repulsion for small r and a weak attraction at large r. The most common phenomenological form of u(r) is the Lennard-Jones or 6-12 potential proposed by John Edward Lennard-Jones in 1924:
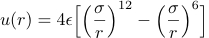 ,
,
The values of σ and ε for argon are σ = 3.4 × 10-10 m and ε = 1.65 × 10-21 J.
The existence of many calculations and simulation results for the Lennard-Jones potential encourages us to consider it even though there are more accurate forms of the interparticle potential for real gas and liquids.
It is possible to simulate a system of Lennard-Jones particles using either molecular dynamics or Monte Carlo methods.
Problems
Updated 28 December 2009.